When a catalyst (such as heat) is added to a system, it usually increases the rate of particle collisions. Solids (bodies made from cohesive elements) have the least kinetic energy, liquids (liquids held together by surface tension) has limited potential kinetic energy, and gases (volume motion without condensation) are the most active. When two particles collide, the faster one will have more kinetic energy than the slower. What Happens to Kinetic Energy When a Catalyst is Added? There are five major forms, including:Įach form has different characteristics but it's all created through kinetic motion- which can be anything from turning on a light, converting chemical energy into electricity, or knocking against something else in order to make noise. Kinetic energy is the type of power that allows for movement. This theory has been an essential part of modern science ever since its inception! What are the Main Types of Kinetic Energy? James Clerk Maxwell & Ludwig Boltzmann, in collaboration with one another, established what is now known as classical thermochemistry. In the 19th century, two great minds led humanity in understanding kinetic energy in Chemistry. This also means that if the temperature is higher, the molecules will move faster and possess greater kinetic energy. This means that if there are more molecules, they will have more kinetic energy. The Kinetic Energy of an object is dependent upon the mass and speed towards which it travels. Kinetic energy refers to the total amount of energy possessed by molecules at a given temperature, not including any potential or gravitational energy.

Atoms or groups of atoms can only absorb specific amounts of energy that are related to their individual structures.The average kinetic energy of a group of gas molecules depends on the temperature.If the collision is perfectly elastic, then the total kinetic energy of the system remains exactly the same. When gas particles collide, they exert equal but opposite forces on each other.Gas particles are in constant motion, colliding with each other continuously.The kinetic energy of molecules increases as temperature rises.
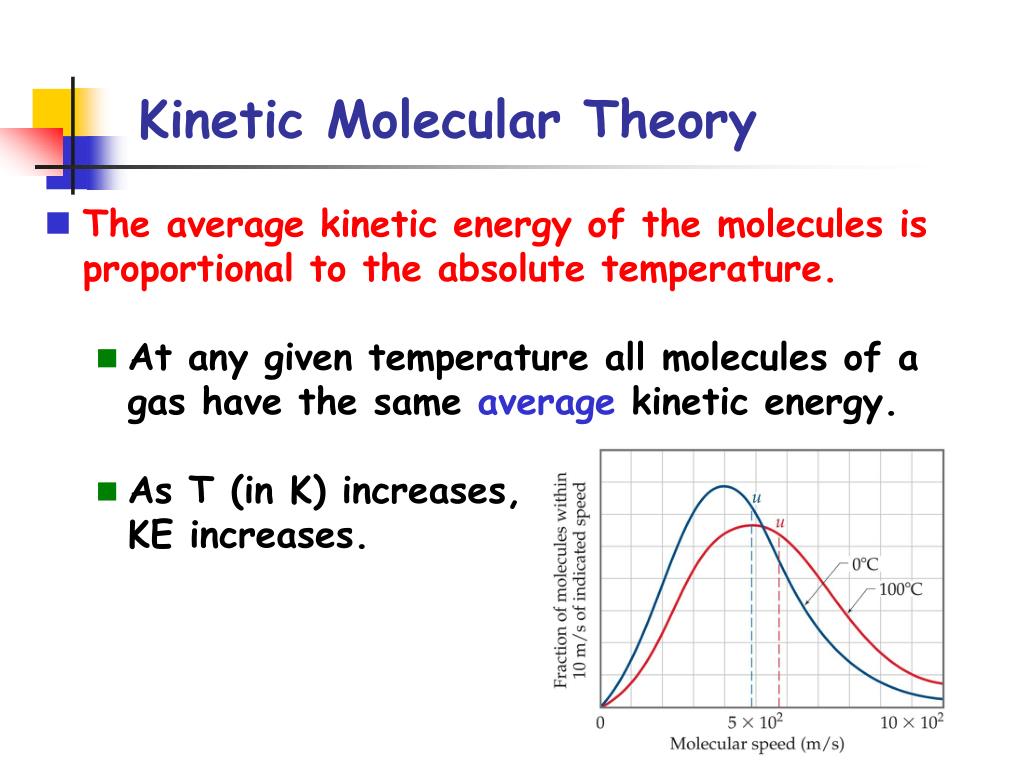
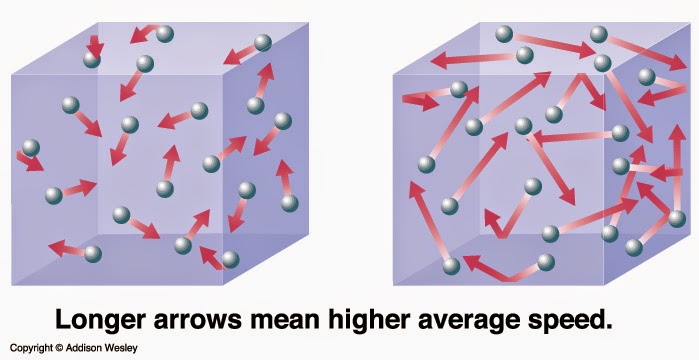
Kinetic Molecular Theory is a model that attempts to explain what happens in terms of groups of atoms and molecules colliding with each other and how those collisions change their energy levels, as well as their physical and chemical properties.
